Using Nanotechnology to Improve Pain and Function with a Novel, Drug-Free, Topical Pain-Relief Patch: An Interim Analysis
Author'(s): Jeffrey A. Gudin1, Derek T. Dietze2, and Peter L. Hurwitz3
1University of Miami School of Medicine, Miami, Florida USA.
2Metrics for Learning LLC, Queen Creek, Arizona, USA.
3Clarity Science LLC, Narragansett, Rhode Island, USA.
*Correspondence:
Peter Hurwitz, Clarity Science LLC, 750 Boston Neck Road, Suite11, Narragansett, RI 02882, USA, Tel +1917 757 0521, Fax +1855-891-8303.
Received: 09 December 2020; Accepted: 27 December 2020
Citation: Gudin JA, Dietze DT, Hurwitz PL. Using Nanotechnology to Improve Pain and Function with a Novel, Drug-Free, Topical Pain-Relief Patch: An Interim Analysis. Anesth Pain Res. 2020; 4(3): 1-10.
Abstract
Rationale: Evidence-based multimodal approaches to pain management include use of combination pharmacotherapy, nonpharmacological, and behavioral interventions. Much focus has been on the use of topically applied analgesics in order to spare the need for oral therapies. Topical analgesics, including pain relief patches, have proven a valuable strategy in the management of mild to moderate levels of pain. Advancements have allowed for the incorporation of nanotechnology into the development of patch therapies, yet there remains a paucity of data surrounding these products.
The purpose of this IRB-approved, minimal risk study was to evaluate patients with mild, moderate, and severe pain and evaluate perceptions of pain treatment with the use of a novel, non-drug, and nanotechnology-based topical pain-relieving patch. An interim analysis was undertaken to screen for unanticipated adverse events and proof of concept for effectiveness.
Methods: This interim analysis evaluated the efficacy of a topical pain-relieving patch (Kailo Pain Patch®, Pain Relief Technologies, USA) in reducing Brief Pain Inventory (BPI) scores in patients experiencing mild, moderate, severe pain. 66 adult patients (39 females, 27 males) with arthritic, neuropathic, or musculoskeletal pain received patches for 30 days. Surveys were administered at baseline, 14-, and 30 days to assess changes in pain severity, pain interference, oral pain medication use, side effects, and satisfaction with patch use.
Results: Treatment group paired data were collected. Mean BPI Severity score decreased 71% (5.2 to 1.5/10;P< .001) and mean BPI Interference score decreased 67% (4.5 to 1.5/10;P< .001) No side effects of treatment were reported. 98% of patients reported “less” or “a lot less” usage of oral medications. 97% of patients were very/extremely satisfied with the patch and preferred the pain-relieving patch to oral medications. Results showed Quality of Life (QoL) improvements in mood, relations with other people, sleep, walking ability, and enjoyment of life.
Conclusions: Interim results indicate that this novel, nanotechnology-formulated topical analgesic pain-relieving patch can reduce BPI pain severity and interference scores and related pain for adult patients with arthritic, neuropathic, and musculoskeletal pain.
Keywords
Introduction
In the United States, pain is the most common reason patients consult primary care providers and a leading cause of disability [1]. Targeted and alternative treatments to address mild, moderate, and chronic pain conditions are needed [2]. The integration of nanotechnology has allowed for the healthcare field to undergo rapid changes that have resulted in new therapies and approaches being approved for use in clinical practice [3]. Researchers have studied the role that nanotechnology may play in influencing nociceptive pathways, including via the use of this technology in topical therapies [2,4]. It is postulated that incorporation of nanomaterials into patch systems allows for the activation or inhibition of ion channels to provide an analgesic effect [5-7].
Current pain relief treatments include medication, physical therapy, surgery, and non-invasive and non-pharmacological approaches, such as cognitive and behavioral therapy [8-11]. Other topical interventions may include topical analgesic pain patches or creams [12,13]. Over the last decade, medications, such as opioids and non-opioid drugs, such as NSAIDS, have significantly increased [14-16]. Many of these medications are associated with bothersome and dangerous side effects, including GI toxicity, addiction tolerance, abuse and even death [5,17-19]. Due to these side effects, there is a focus on identifying targeted, non-invasive multi-modal approaches that minimize the reliance on pharmaceutical agents- leading to a reduction in side effects and improved quality of life [20].
Novel advancements in nanotechnology and patch delivery systems have the potential to address chronic pain, reduce patient suffering, and improve outcomes [2].
Nanotechnology incorporates materials at a nanometer scale and allows for the enhancement of several classes of materials with enhanced properties. It is known that nanomaterials have unique electrical, chemical, and magnetic properties that may present advantages over traditional materials [21,22]. Research has shown that certain metals can be utilized in various therapeutic applications and at a nanoparticle level with success, and nanotechnology has been studied to address various pain conditions [2,21,23]. For perspective, a nanometer is one billionth of a meter and several orders of magnitude smaller than a micron, approximately the size of a molecule itself. These advances allow for the interaction with cells and tissues at a molecular level with a high degree of functional specificity [24].
Action potentials, which assist with signaling in the brain, rely on the proper functioning of ion channels [25]. There has been an increase in our understanding of the flow of ions within a neuronal circuit and the various signaling components contained within them [26]. It is well known that influencing the body’s electromagnetic field may elicit changes in the voltage-gated calcium channels (VGCC) and voltage-gated ion channels (VGIC) within cells [27]. Published research has demonstrated that alternative therapies incorporating electromagnetic therapy have emerged as a safe and effective option for chronic pain patients across various clinical settings [28].
Current pain guidelines encourage a multi-modal approach to managing pain that includes topical, non-invasive, and non- pharmacological approaches [29]. This approach has been shown safe and effective for chronic pain patients and has the potential to minimize side effects associated with traditional unimodal oral therapy. Research has shown that topical analgesic treatment can reduce pain, improve function, and improve quality of life [12,13].
This Pain Relief: Experiencing and Validity: Evaluating NanoTechnology (PREVENT) study evaluated patients with mild, moderate, severe, and chronic pain and their overall perceptions of pain treatment and associated symptoms with the use of a proprietary non-invasive and non-drug topical patch, the Kailo Pain Patch® (Pain Relief Technologies, USA). The 2.75 x 5.5- inch adhesive pain patch contains copper, silver, and silicon and is printed on a laminate film. Through the combination of the conductive and semi-conductive material, the patch is designed to provide analgesia through nano-capacitors that emit and absorb electromagnetic radiation/energy, thereby influencing the ion signals sent to and received from the nervous system.
An interim analysis of the study data was undertaken to screen for unanticipated adverse events and proof of concept for effectiveness. The analysis assessed changes in Brief Pain Inventory short form (BPI) pain severity and pain interference scores, as well as a change in the use of pain medications 14- and 30-days following treatment. Future analyses will include reporting differences between the treatment group, control group, and also in a control- crossover group of patients (after 30 days in the control group).
Methods Study Design
This study is a prospective, Institutional Review Board-approved Observational Study aimed at evaluating patients’ experiences and/or perceptions and analgesic response for those who have received from their treating clinician a non-pharmacological, nanotechnology-formulated OTC topical pain-relieving patch (Kailo Pain Patch®, Pain Relief Technologies LLC, Sandy, Utah, USA). This interim analysis evaluated patient answers to baseline, day 14-, and day 30 surveys from validated pain measurement and symptom scales (e.g., Brief Pain Inventory (BPI)) as well as additional survey questions regarding pain medication use, patient satisfaction, patient quality of life, and resumption of their normal activities. Future analysis will include the evaluation of a Control Group of patients that were not prescribed a Kailo Pain Patch® at enrollment, and a cross- over group patient who received the Kailo Pain Patch® after 30 days of being in the control group.
The study protocol was approved by IntegReview institutional review board and was performed in full accordance with the rules of the Health Insurance Portability and Accountability Act of 1996 (HIPAA) and the principles of the declaration of Helsinki and the international council of Harmonisation/GCP. All patients gave informed and written consent. Patients who met the eligibility criteria and who were treated with the pain-relieving patch comprised the study’s treatment group (TG). Patient answers to surveys were used to evaluate pain relief by comparing answers to validated pain measurement scales (e.g., BPI) as well as other survey questions including patient satisfaction.
Topical Intervention
This nanotechnology-formulated OTC topical pain-relieving patch (the Kailo Pain Patch®, Pain Relief Technologies LLC, Sandy, Utah, USA) contains a composite of 2 conductive elements (copper and silver) and 1 semi-conductive element (silicon). Patients in the treatment group were instructed to wear their patch as needed and for as long as needed (Picture 1 and 2).
Patients
For the treatment group, patient inclusion criteria were as follows: 1) ages 18 to 65 years, inclusive; 2) ability to provide written informed consent; 3) received a non-pharmacological, nanotechnology formulated topical pain-relieving patch from their treating clinician; 4) had been diagnosed with a mild, moderate, or severe pain condition. Patients who had had a history of use drug or alcohol abuse, patients who had an implantable pacemaker or defibrillator, or patients who were pregnant, were ineligible to participate in the study.
Each site provided patients an identification number to keep personal information confidential in compliance with the HIPAA, and a confidential file containing the informed consent forms and patient identification numbers were kept and maintained in a secured cabinet only accessible to the principal investigator and authorized personnel. Patient survey responses were provided to Clarity Science LLC with no identifying information about patients.
Patients could withdraw from this study at any time with the assurance of no harmful or unfavorable impact on their medical care. All diagnostic tests and treatment decisions were made at the discretion of clinicians, with no tests, treatments, or investigations performed as part of this study.
Study procedures and assessments
At the time of enrollment, patients were asked to complete a baseline survey and were then asked to complete follow-up surveys on days 14 and 30 of the study period. The baseline and follow-up surveys are comprised of questions to address the primary pain complaint of the patient, including: 1) arthritis; 2) neuropathy or radiculopathy; 3) myofascial or musculoskeletal pain or spasm; or 4) other. Once patients chose their primary pain complaint, and confirmed by the clinician, they indicated the location of that pain in areas such as hands, feet, hips, knees, neck, shoulders, and back, among others. Patients indicated only one pain complaint and location of pain for which the patch was primarily being used.
Patients completed the BPI as part of each survey. The BPI is often used as a measure of pain for a wide range of conditions including cancer, musculoskeletal disorders, depressive conditions, and surgical pain. The BPI is commonly recommended for use in clinical trials of patients with chronic pain and has adequate internal consistency, acceptable-to-excellent test-retest reliability; satisfactory-to-good construct validity, criterion validity, and is sensitive to change [30-34]. Ratings on the BPI are based on a 0-10 numerical scale. For the questions about pain severity, 0 is “no pain” and 10 is “pain as bad as you can imagine.” For the questions about pain interference with activities of daily living, 0 is “does not interfere” and 10 is “completely interferes.” Patient responses to questions regarding pain severity (4 questions) and pain interference (7 questions) were compiled to yield the overall score for pain severity and pain interference.
Patients were asked to indicate any other medications that they had been taking for pain relief at the time of the baseline, day 14, and day 30. Categories of medications that patients could choose included OTC agents (e.g., ibuprofen, naproxen, acetaminophen, aspirin, and other pain medications such as creams, gels, roll-ons, sprays, patches or rubs), prescription NSAIDs, prescription opioids, skeletal muscle relaxants, (e.g., methocarbamol, cyclobenzaprine, metaxalone) or prescription anticonvulsants (e.g., gabapentin or pregabalin). Patients could indicate use of more than one type/ class of analgesic medication.
Study end points
There were four primary endpoints:
- Changes in patient Brief Pain Inventory (BPI) overall severity and interference scores among and between the treated groups and the control groups for the primary pain complaint.
- Changes in the use of prescription and OTC medications (other than Kailo Pain Patch®).
- Demographics and patient satisfaction with the Kailo Pain Patch® treatment.
- Any side effects reported by patients which are summarized using descriptive statistics
Statistical analysis
For all variables, descriptive statistics were calculated, including frequencies and percent for categorical variables and means with standard deviation (SD) for continuous variables. The maximum sample size available was used for each statistical analysis.
For this interim report, changes from baseline to day 14, and to day 30, in BPI mean pain severity and pain interference scores were analyzed using the paired t test to identify any statistically significant differences within the treatment group. Each survey collected the numbers and types of prescription and OTC oral/ topical medications being used for pain relief; statistically significant differences in the use of these types of medications from baseline to day 30 were determined using the McNemar test and X 2 test for binomial paired and unpaired data respectively. Descriptive statistics were used to determine patient satisfaction with the pain-relieving patch within those treated. Descriptive statistics were also used to report any side effects experienced by patients.
A two-tailed alpha was set to 0.05 for all statistical comparisons. SPSS v. 27 was used for all analyses.
Results
Baseline demographic and clinical characteristics of patients
A total of 66 patients at 2 US investigator sites were enrolled in the study and completed the baseline, day 14 and day 30 surveys.
Demographic results were similar for gender and age at the baseline survey for this interim analysis of treated patients. Of the 66 patients, 27 (40.9%) were male and 39 (59.1%) were female. The mean age at baseline was 48.0 years.
The primary pain complaint for these treated patients was recorded at baseline (Table 1). Myofascial/musculoskeletal pain was the most prominent pain complaint indicated by 43/66 (65%) of patients. Neuropathy/radiculopathy was the next most common pain complaint for 12/66 (18%) of patients, and arthritis was the third most prominent primary complaint indicated by 11/66 (17%) of patients.
At baseline, study participants who indicated myofascial/ musculoskeletal pain as their primary complaint noted that their back and lower extremities was the most common location of pain (n=32). Patients who indicated neuropathy/radiculopathy as their primary pain complaint noted their back was the most common location of their pain (n=9).
Over 88% of patients (59/66) indicated that they had their pain for over 3 months. Almost 32% of patients reported having their pain for 3 months to one year (21/66) and over 57% reported having pain for more than one year (38/66).
Baseline BPI severity and interference scores
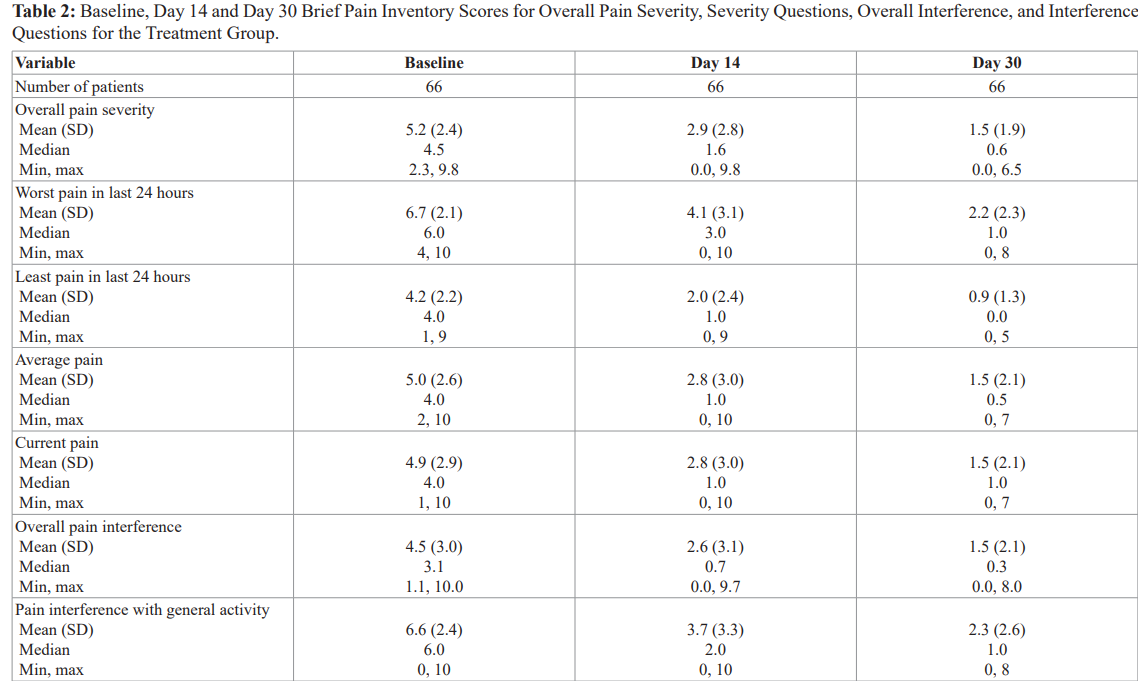
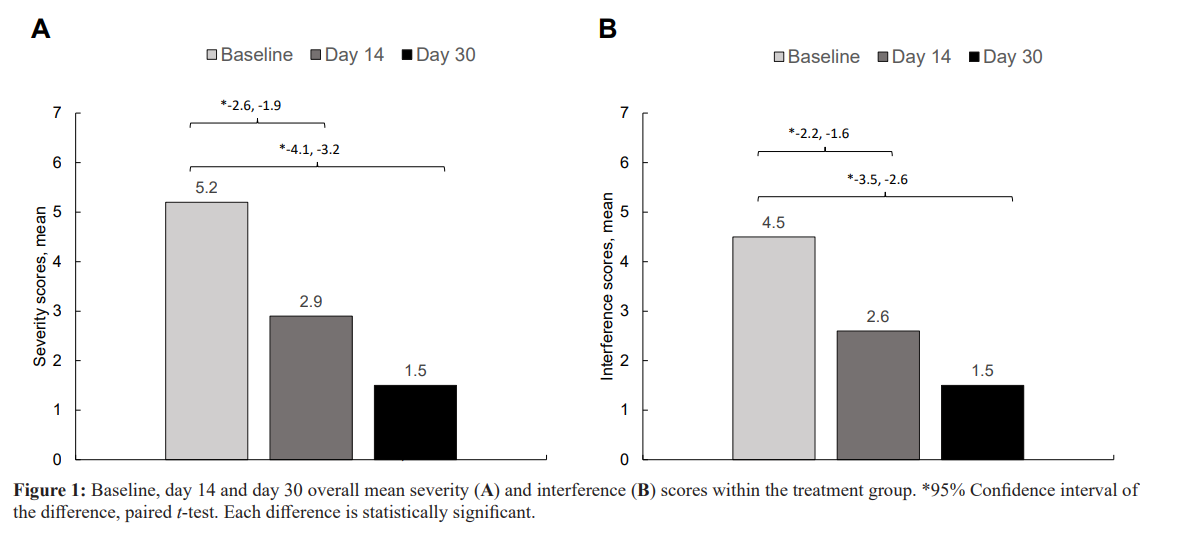
The mean BPI pain severity score at baseline was 5.2, with SD = 2.4 (Table 2). The baseline mean BPI interference score was 4.5, with SD = 3.0.
Changes from baseline to day 14 and day 30 in mean BPI pain severity scores
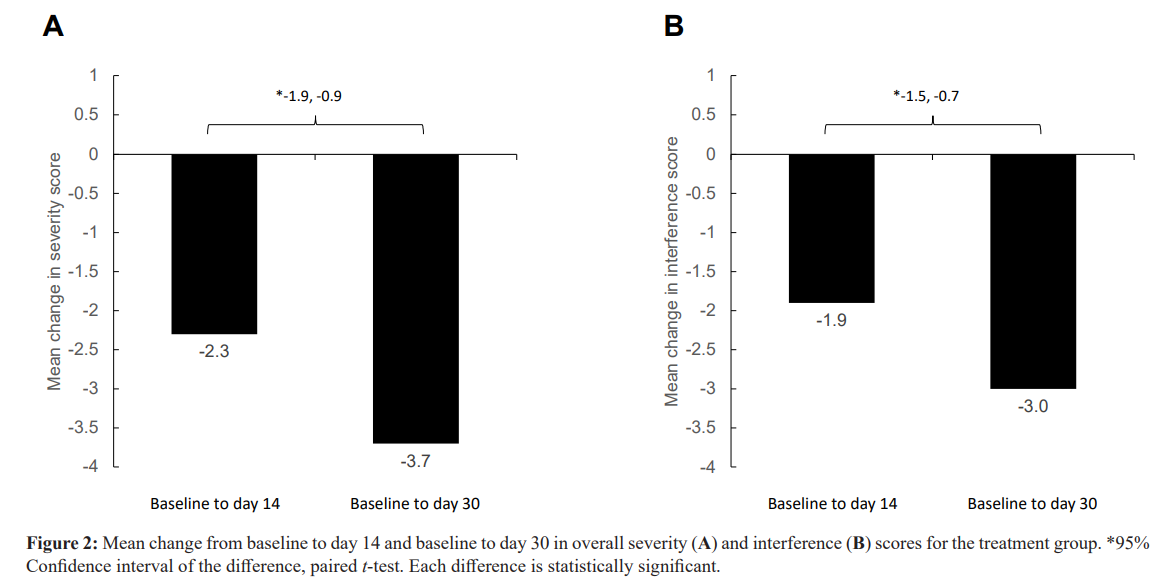
Patients showed a 44% decrease (5.2 to 2.9, 95% CI, -2.6 to -1.9, p < 001) from baseline to day 14 and a 71% decrease (5.2 to 1.5, 95% CI, -4.1 to -3.2, p < 001) from baseline to day 30 in mean BPI severity scores after using the Kailo Pain Patch (Figure 1A). For each of the 4 questions which comprise the BPI pain severity score (pain at its worst and least in the last 24 hours, pain right now, and how much pain on average), see Table 2, changes in mean scores from baseline to day 14 and to day 30 all decreased statistically significantly (p < 001). The amount of decrease from baseline to day 30 was greater than the decrease from baseline to day 14 for each of the 4 questions.
Changes from baseline to day 14 and day 30 in mean BPI pain interference scores
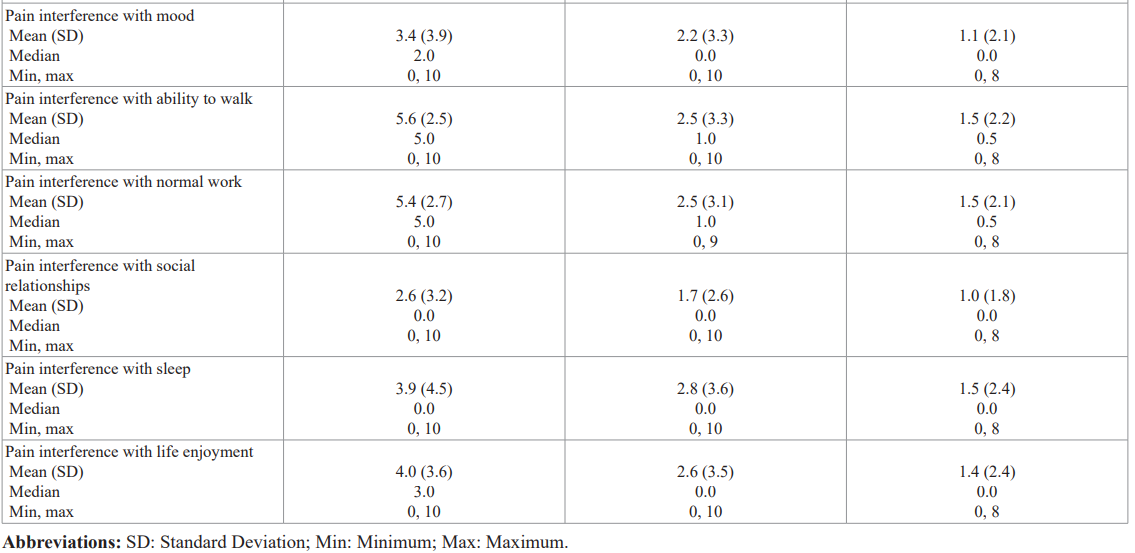
In the TG, all 7 ratings of BPI pain interference scores (general activity, mood, walking ability, normal work, relations with other people, sleep, and enjoyment of life) decreased statistically significantly (p< 001), from baseline to day 14 and day 30 (Figure 1B). At day 14, patients had a 42% decrease (4.5 to 2.6, 95% CI, -2.2 to -1.6, p < 001) that improved to a 67% decrease (4.5 to 1.5, 95% CI, -3.5 to -2.6, p < 001) at day 30. The amount of decrease in mean pain interference scores from baseline to day 30 was statistically significantly greater than from baseline to day 14 (95% CI, -1.5 to -0.7, p < 001) (Figure 2B).
Changes in self-perceived pain relief from medications
One of the BPI questions (not part of the pain severity or interference scores) asks the patient how much pain relief (in increments of 10% from 0% = no relief to 100% = complete relief) they have experienced from treatments or medications within the last 24 hours. At baseline, patients reported a mean of 24% pain relief from current treatment or medications; by day 14 they reported 75% pain relief, and by day 30 they reported 86% pain relief. The change in mean percent relief from baseline to day 14 was statistically significant (95% CI, 43.1 to 58.4, p < 001) and was also significant from baseline to day 30 (95% CI, 56.9 to 67.5,p < 001).
In each survey, patients indicated the type of medication they were taking for pain relief including OTC pain medications, prescription anti-inflammatory medications, opioids or anticonvulsants, or muscle relaxants. At baseline, there were 62/66 (94%) patients taking at least one medication. At day 14, that number had decreased by 59% to 26/66 (39%) patients taking at least one medication (p < 001, McNemar test), and at day 30, that number remained the same (26/66, 39%, p < 001).
The total number of patients taking OTC pain medication at baseline was 29/66 (44%) (Figure 3). There was no statistically significant change in use of OTC pain medications from baseline to day 14 or from baseline to day 30 (p = .125 for each, McNemar test). Ibuprofen was the highest reported OTC pain medication in use at baseline (19/66, 29%). Acetaminophen was the second most common OTC pain medication used by 18/66 (27%) of patients. As far as prescription anti-inflammatory medication, diclofenac
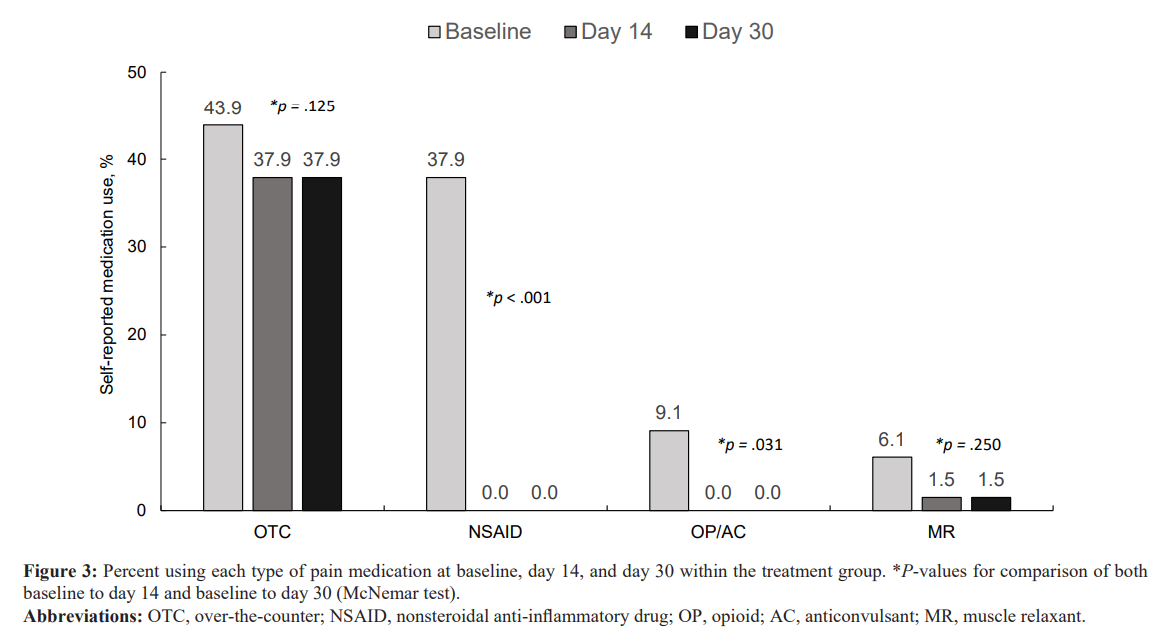
was reported most often 15/66 (23%). All treated patients discontinued their prescription NSAID by day 14 which persisted through day 30 (a statistically significant decrease of p < 001 for each, McNemar test).
Although a minority of patients reported using opioids or anticonvulsants at baseline 6/66 (9%), all treated patients discontinued their prescription opioids and anticonvulsants by day 14 which persisted through day 30 (a statistically significant decrease of p = .031 for each, McNemar test).
Separate from indicating use of specific medications for pain, patients were asked how their use of oral pain medications had changed (scale: 1 = A lot more, 2 = More, 3 = No change, 4 = Less, 5 = A lot less). At day 14, the mean rating was 4.5 (SD = 0.85, n = 65) with 91% reporting “less” or “a lot less.” At day 30, the mean rating was 4.8 (SD = 0.4, n = 65) with 99% reporting “less” or “a lot less.”
Satisfaction with use of the pain patch
Day 14 satisfaction ratings related to specific aspects of use of the pain relief patch (scale: 1 = strongly disagree, 2 = Disagree, 3 = Neutral, 4 = Agree, 5 = strongly agree) were 4.8 for each of “easy to apply” and “convenient,” 4.6 for “preferred over other topical pain-relieving treatments,” and 4.5 for “preferred over pills/oral medication.” At day 30, the mean ratings were 4.9 for each of “easy to apply” and “convenient,” and 4.8 for each of “preferred over pills/oral medication” and “preferred over other pain-relieving treatments.” At day 30, overall satisfaction was 4.8 out of 5 (scale: 1 = not at all, 2 = Not very, 3 = somewhat, 4 = Very, 5 = extremely).
Use of the Patch
At day 14, 61/66 (92%) of patients reported that they kept the patch on ‘ almost all of the time.’ The remaining 5/66 (8%) of patients reported that they used the patch ‘ until the pain was gone, then again when the pain came back.’ At day 30, 49/66 (74%) of patients reported that they kept the patch on ‘ almost all of the time.’ The remaining 17/66 (26%) of patients reported that they used the patch ‘ until the pain was gone, then again when the pain came back.’
At the first follow-up data collection point at day 14, 40/66 (61%) of patients reported that they felt pain relief in less than 5 minutes after applying the pain relief patch. 38% of patients (25/66) reported that it took longer than 10 minutes to feel pain relief. At day 30, 55/66 (83%) of patients reported that they felt pain relief in less than 5 minutes after application and 10/66 (15%) of patients reported pain relief after 10 minutes. At day 14, out of the 25 patients for whom it took longer than 10 minutes to achieve pain relief after initial application, 18/25 (72%) readjusted the location of their patch. At day 30, out of the 10 patients who did not experience pain relief within 10 minutes, 6/10 (60%) reported readjusting the location of the patch. On average, at day 14, 41/66 (62%) of patients reported attempting only one location before experiencing pain relief and 22/66 (33%) of patients reported attempting 2 locations before they experienced pain relief. Three patients reported attempting more than 3 locations before finding pain relief. At day 30, 57/66 (86%) of patients reported attempting one location before experiencing pain relief and 9/66 (14%) attempted 2 locations prior to experiencing pain relief. Only 1/66 patients reported no pain relief, despite attempts at trying to readjust the location of the patch.
Duration of Pain Relief
At day 30, patients were asked how long it took for the pain to return once they removed the patch. 27% of patients (18/66) reported that their pain did not return after they removed the patch; further follow up was not planned for this study. 55% of patients (36/66) reported that it took longer than one day for the pain to return after patch removal, and 12/66 (18%) of patients reported that pain returned under 2 hours after the pain patch was removed.
Safety
Patients reported no adverse skin reactions, adverse or serious adverse events while being treated with the pain relief patch.
Safety
Patients reported no adverse skin reactions, adverse or serious adverse events while being treated with the pain relief patch.
Discussion
Here we report interim results of the PREVENT study, a prospective, non-randomized study of patients presenting with mild, moderate, or severe arthritic, neurological, and musculoskeletal pain. Patients indicated their utilization of pharmacological treatments for pain at baseline, 14 days, and 30 days. Treatments included OTC agents, prescription NSAIDs, opioids, anticonvulsants, or a combination of those four classes. BPI scores indicated that patients in the treatment group were experiencing moderate or severe pain.
In this interim analysis, changes in BPI pain severity and pain interference scores and use of concurrent pain medications from baseline to day 14, and to day 30, were evaluated to assess the safety and efficacy of the Kailo Pain Patch® for the treatment of pain reported by enrolled patients. Limited data were available for patients enrolled in the control group and subsequent crossover group, and therefore these data are not reported here. Future data assessments will include comparisons of all patient groups, and outcomes of these results will be reported. This interim analysis showed that patients reported positive results after utilizing the nanotechnology-formulated pain patch that led to a statistically significant reduction in mean BPI pain severity and interference score. There were no side effects reported with the topical pain- relieving patch.
Understanding the pathophysiology of pain may help to explain the preliminary results above. Noxious stimuli are transduced into electrical signals in free “unencapsulated” nerve endings that have branched from the main axon and terminate in the wall of arterioles and surrounding connective tissue, and may innervate distinct regions in the dermis and epidermis [35]. It is theorized that the mechanisms of analgesic effect come from the discharges of billions of capacitors contained in the Kailo Pain Patch® when encountering the radiation of ambient energy emitted from the body’s natural or nociceptive-related electrical charges. As electrical signals are sent along the body’s nervous system pathway, that electrical potential is large enough to be sensed by these capacitors. By placing the Kailo Pain Patch® along the nociceptive pathway, it is thought to attract the signal where poor function is occurring, charging the capacitors and then influencing the axons along the body’s pathway and acting as a ‘ bridge’ over the portion of the pain pathway where electrical disruption is occurring. It is known that with advancing age in in cases of injury, the body’s ability to transmit proper signals along the nervous system pathway is negatively affected [36]. The Kailo Pain Patch® is theorized to be clarifying or enhancing the signal and communication along the pain pathway and nervous system and not blocking the pain pathways. This, in part, is thought to be due to some patients experiencing long term pain reduction after the patch is removed where pain does not immediately return after patch removal. In some cases, it could also potentially be due to promoting the body’s natural healing process.
There is an unmet need for alternative treatment options for patients with pain. Considering the bothersome and dangerous adverse effects that occur with NSAIDS, acetaminophen, opioids and adjuvant analgesics, the incorporation of novel non- pharmacological treatment options that include nanotechnology will add important safe and effective treatment options. Reductions in addiction, abuse, GI and other systemic side effects, toxicity issues, cardiovascular and renal implications may be able to be lessened thorough the use of these non-pharmacological approaches [37-42].
There is an unmet need for alternative treatment options for patients with pain. Considering the bothersome and dangerous adverse effects that occur with NSAIDS, acetaminophen, opioids and adjuvant analgesics, the incorporation of novel non- pharmacological treatment options that include nanotechnology will add important safe and effective treatment options. Reductions in addiction, abuse, GI and other systemic side effects, toxicity issues, cardiovascular and renal implications may be able to be lessened thorough the use of these non-pharmacological approaches [37-42].
Interim results from this IRB-approved observational study suggests that the non-pharmacological and nanotechnology formulated pain patch studied may provide a viable treatment alternative to pharmacological based therapies. Further analysis and formal clinical controlled trials are planned to confirm these results.
Limitations
This was an observational study based on a sample of patients attending diverse clinical settings for the treatment of arthritic, neurological, and musculoskeletal pain who consented to participate in this study. This interim analysis reported only on the group of patients who were treated with the Kailo Pain Patch®. In future analyses, control group and control-crossover group patients will be evaluated. There were limited numbers of those groups to conduct a meaningful analysis at this time. Generalizability of the findings may be limited to the TG only because those patients received topical therapies.
The data of those patients who did not complete the follow up surveys after baseline were removed from evaluation. Although patients were given instruction on proper patch placement and timing of use, patients individually decided patch placement and used the patch on an ‘ as needed’ basis. This may limit the consistency in patch utilization and overall results among all patient groups as each patient utilized the patch according to their own needs. Although the FDA has provided guidance of the value of incorporating and reporting on this type of Real-World Evidence data, in addition to utilizing validated pain scales for data collection, there may be a limitation on the interpretability and validity of these results due to lack of consistency among patient data as is collected in a randomized and formal clinical trial [43].
Conclusion
These interim results suggest that the non-pharmacological topical pain patch studied was effective and safe for the relief of moderate and severe pain attributed to arthritic, neurological, and musculoskeletal disorders. Significant reductions in pain were reported, which supports the use of novel nanotechnology systems into the treatment algorithm for a multi-modal and opioid sparing pain management approach. These interim results support the use of this OTC pain patch as a first-line non-pharmacological treatment option and should be considered by healthcare professionals and patients.
Disclosure
Jeffrey A Gudin has received compensation from Clarity Science LLC for his role as principal investigator and for providing protocol- required services for the study. Peter L Hurwitz is President of Clarity Science LLC. Derek T Dietze received compensation for study statistical analyses.
Acknowledgments
This IRB-approved study administered by Clarity Science LLC was funded by Pain Relief Technologies, the distributors of the Kailo Pain Patch®.
References
1.Finley CR, Chan DS, Garrison S, et al. What are the most common conditions in primary care? Systematic review. Can Fam Physician. 2018; 64: 832-840.
2.Chen J, Jin T, Zhang H. Nanotechnology in Chronic Pain Relief. Front Bioeng Biotechnol. 2020; 8: 682.
3.Ventola CL. Progress in nanomedicine: approved and investigational nanodrugs. Pharmacy Therapeutics. 2017; 42:742-745.
4.Kumar A, Tan A, Wong J, et al. Nanotechnology for Neuroscience: Promising Approaches for Diagnostics, Therapeutics and Brain Activity Mapping. Adv Funct Mater. 2017; 27: 1700489.
5.Zhuo M. Neural mechanisms underlying anxiety–chronic pain interactions. Trends Neurosci. 2016; 39: 136-145.
6.Tang Z, Zhao P, Ni D, et al. Pyroelectric nanoplatform for NIR-II-triggered photothermal therapy with simultaneous pyroelectric dynamic therapy. Mat. Horiz. 2018; 5: 946-952.
7.Zhang C, Bu W, Ni D, et al. Synthesis of iron nanometallic glasses and their application in cancer therapy by a localized Fenton reaction. Angew Chem Int Ed. 2016; 55: 2101-2106.
8.Volkow N, Benveniste H, McLellan AT. Use and misuse of opioids in chronic pain. Ann Rev Med. 2018; 69: 451-465.
9.Ambrose KR, Golightly YM. Physical exercise as non- pharmacological treatment of chronic pain: why and when. Best Pract Res Clin Rheumatol. 2015; 29: 120-130.
10.Ducic I, Mesbahi AN, Attinger CE, et al. The role of peripheral nerve surgery in the treatment of chronic pain associated with amputation stumps. Plastic Reconst Surgery. 2008; 121: 908-914.
11.De Williams AC, Eccleston C, Morley S. Psychological therapies for the management of chronic pain (excluding headache) in adults. Cochr Data Syst Rev. 2012; 11: 407.
12.Gudin JA, Brennan MJ, Harris ED, et al. Changes in pain and concurrent pain medication use following compounded topical analgesic treatment for chronic pain: 3- and 6-month follow- up results from the prospective, observational Optimizing Patient Experience and Response to Topical Analgesics study. J Pain Res. 2017; 10: 2341-2354.
13.Gudin JA, Dietze DT, Hurwitz PL. Improvement of Pain and Function. After Use of a Topical Pain Relieving Patch: Results of the RELIEF Study. J Pain Res. 2020; 13: 1557-1568.
14.Sullivan MD, Howe CQ. Opioid therapy for chronic pain in the United States: promises and perils. PAIN. 2013; 154: 94- 100.
15.Ballantyne JC, Sullivan MD. Intensity of chronic pain—the wrong metric. N Engl J Med. 2015; 373: 2098-2099.
16.Kaye AD, Cornett EM, Hart B, et al. Novel pharmacological nonopioid therapies in chronic pain. Curr Pain Head Rep. 2018; 22: 31.
17.Pohl M, Smith L. Chronic pain and addiction: challenging co- occurring disorders. J Psych Drugs. 2012; 44: 119-124.
18.Vowles KE, McEntee ML, Julnes PS, et al. Rates of opioid misuse, abuse, and addiction in chronic pain: a systematic review and data synthesis. Pain. 2015; 156: 569-576.
19.Volkow ND, McLellan AT. Opioid abuse in chronic pain— misconceptions and mitigation strategies. New Eng J Med. 2016; 374: 1253–1263.
20.Gao YJ, Ji RR. Targeting astrocyte signaling for chronic pain. Neurotherapeutics. 2010; 7: 482-493.
21.Bleeker EAJ, de Jong WH, Geertsma RE, et al. Considerations on the EU definition of a nanomaterial: science to support policy making. Regul Toxicol Pharmacol. 2013; 65: 119-125.
22.Taniguchi N, “On the basic concept of ‘nano-technology’”, presented at Proc. Int. Conf. Prod. Eng. Tokyo, Part II, Tokyo, 1974.
23.Siavash Beiranvand, Mohamad Masud Sorori. Pain management using nanotechnology approaches, Artificial Cells, Nanomedicine and Biotechnology. 2019; 47: 462-468.
24.Siddhartha Shrivastava, Debabrata Dash. Applying Nanotechnology to Human Health: Revolution in Biomedical Sciences, Journal of Nanotechnology. 2009; 184702.
25.Spillane J, Kullmann DM, Hanna MG. J Neurol. Neurosurg Psychiatry. 2016; 87: 37.
26.Pancrazio JJ. Nanomedicine. 2008; 3: 823.
27.Pall ML. Electromagnetic fields act via activation of voltage- gated calcium channels to produce beneficial or adverse effects. J Cell Mol Med. 2013; 17: 958-965.
28.Arneja AS, Kotowich A, Staley D, et al. Electromagnetic fields in the treatment of chronic lower back pain in patients with degenerative disc disease. Future Sci OA. 2016; 2:105.
29.Cuomo A, Bimonte S, Forte CA, et al. Multimodal approaches and tailored therapies for pain management: the trolley analgesic model. J Pain Res. 2019; 12: 711-714.
30.Dworkin RH, Turk DC, Wyrwich KW, et al. Interpreting the clinical importance of treatment outcomes in chronic pain clinical trials: IMMPACT recommendations. J Pain. 2008; 78: 1155-1162.
31.Erdemoglu AK, Koc R. Brief pain inventory score identifying and discriminating neuropathic and nociceptive pain. Acta Neurol Scand. 2013; 128: 351-358.
32.Keller S, Bann CM, Dodd SL, et al. Validity of the brief pain inventory for use in documenting the outcomes of patients with noncancer pain. Clin J Pain. 2004; 20: 309-318.
33.Mendoza T, Mayne T, Rublee D, et al. Reliability and validity of a modified brief pain inventory short form in patients with osteoarthritis. Eur J Pain. 2006; 10: 353-361.
34.Tan G, Jensen MP, Thornby JI, et al. Validation of the brief pain inventory for chronic nonmalignant pain. J Pain. 2004; 5: 133-137.
35.Dubin AE, Patapoutian A. Nociceptors: the sensors of the pain pathway. J Clin Invest. 2010; 120: 3760-3772.
36.Yezierski RP. The effects of age on pain sensitivity: preclinical studies. Pain Med. 2012; 13: 27-36.
37.Farkouh ME, Greenberg BP. An evidence-based review of the cardiovascular risks of nonsteroidal anti-inflammatory drugs. Am J Cardiol. 2009; 103: 1227-1237.
38.Harirforoosh S, Jamali F. Renal adverse effects of nonsteroidal anti-inflammatory drugs. Expert Opin Drug Saf. 2009; 8: 669-681.
39.John R, Herzenberg AM. Renal toxicity of therapeutic drugs. J Clin Pathol. 2009; 62: 505-515.
40.Lazzaroni M, Porro GB. Management of NSAID-induced gastrointestinal toxicity: focus on proton pump inhibitors. Drugs. 2009; 69: 51-69.
41.Scarpignato C, Hunt RH. Nonsteroidal anti-inflammatory drug-related injury to the gastrointestinal tract: clinical picture, pathogenesis, and prevention. Gastroenterol Clin North Am. 2010; 39: 433-464.
42.Trelle S, Reichenbach S, Wandel S, et al. Cardiovascular safety of non-steroidal anti-inflammatory drugs: network meta-analysis. BMJ. 2011; 342: 7086.
43.Upton FHR 6—21st century cures act. 114th congress (2015– 2016) Jul 13, 2015. Available from: www.congress.gov/ bill/114th-congress/house-bill/6/text. Accessed January16, 2018.